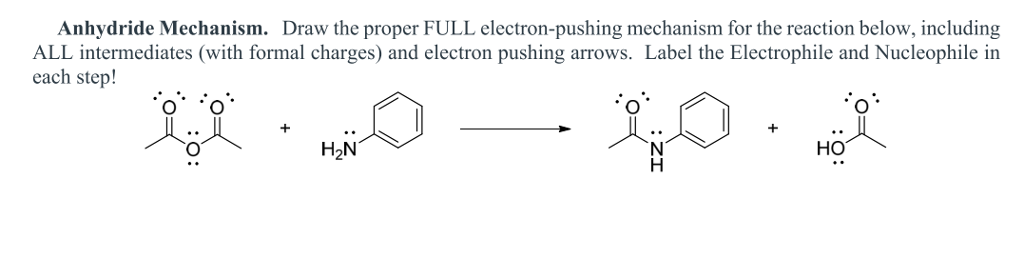
The most reactive of the carboxylic acid derivatives frequently found in biomolecules are the acyl phosphates. Ch21 Carboxylic acid Derivatives(landscape). Amides do undergo acyl substitution reactions in biochemical pathways, but these reactions are inherently slow and the enzymes catalyzing them have evolved efficient strategies to lower the activation energy barrier.Ĭarboxylic acids and esters are in the middle range of reactivity, while thioesters are somewhat more reactive. The reactivity depends on both the structure of the derivative and also on the nature of the nucleophile. 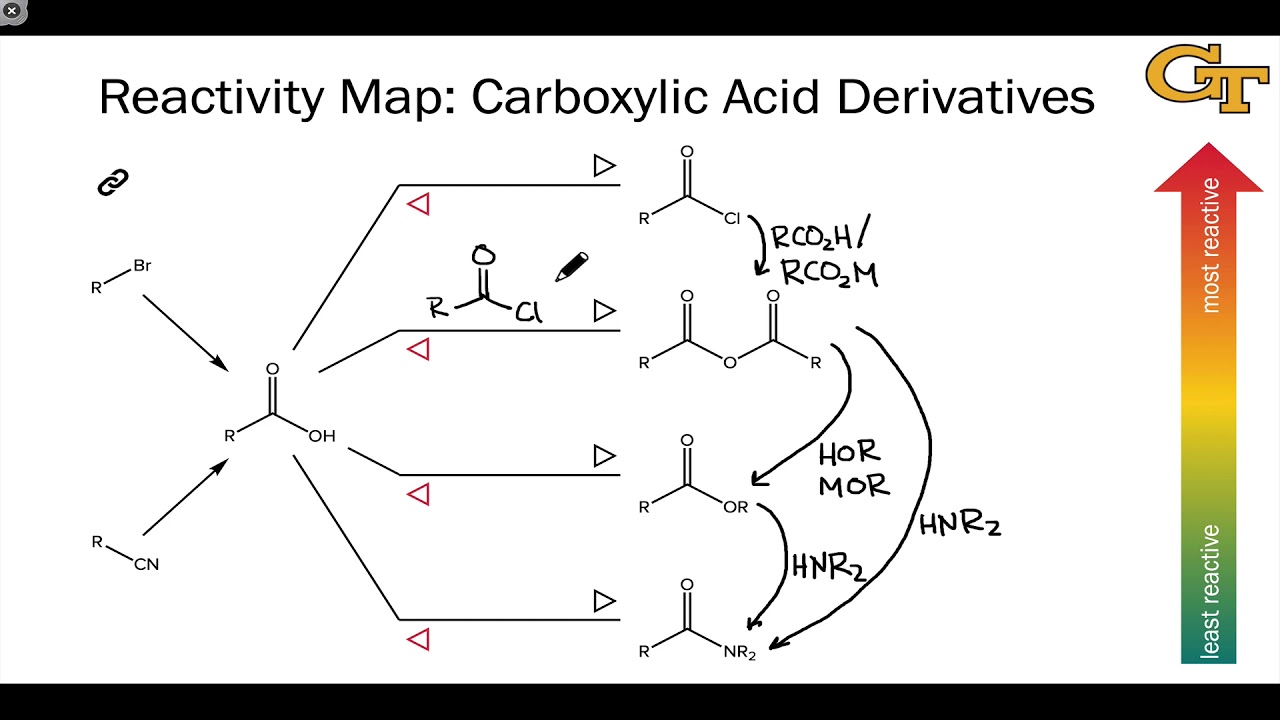
In amides, the nitrogen atom is a powerful electron donating group by resonance – recall that the carbon-nitrogen bond in peptides has substantial double-bond character – thus amides are relatively unreactive. The more electronegative leaving groups withdrawn electron density from the carbonyl, thereby, increasing its electrophilicity. The negatively charged oxygen on the carboxylate group has lots of electron density to donate, thus the carbonyl carbon is not very electrophilic. The relative reactivity of carboxylic acid derivatives toward nucleophile substitutions is related to the electronegative leaving group’s ability to activate the carbonyl. 5) Understand that the reactivity of carboxylic acid derivatives depends on 2 things. Some examples of these functional derivatives were displayed earlier. Carboxylic Acids & Carboxylic Acid Derivatives. The reactivity of acid derivatives can be correlated to the leaving group ability of the base that is expelled. Following this mechanism, it is possible to transform one acid derivative into another one. The general mechanism of this process is given below. This depends on how much electron density the neighboring heteroatom on the acyl X group is able to donate: greater electron donation by the heteroatom implies lower partial positive charge on the carbonyl carbon, which in turn implies lower electrophilicity. Different carboxylic acid derivatives have very different reactivities, acyl chlorides and bromides being the most reactive and amides the least reactive, as. Carboxylic acids have a hydroxyl group bonded to an acyl group, and their functional derivatives are prepared by replacement of the hydroxyl group with substituents, such as halo, alkoxyl, amino and acyloxy. (21-5) Acid derivatives normally react by nucleophilic acyl substitution. Here’s another way to think about the relative reactivites of the different carboxylic acid derivatives: consider the relative electrophilicity, or degree of partial positive charge, on the carbonyl carbon in each species. This is why it is not possible to directly convert an ester, for example, into a thioester by an acyl substitution reaction – this would be an uphill reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
